- Research article
- Open access
- Published:
Lead optimization for promising monoamine oxidase inhibitor from eugenol for the treatment of neurological disorder: synthesis and in silico based study
BMC Chemistry volume 13, Article number: 38 (2019)
Abstract
Natural based inhibitors of monoamine oxidase are promising drug candidates for the treatment of several neurodegenerative and neuropsychological disorders including depression, anxiety, Parkinson’s disease and Alzheimer’s disease. In the present study we designed and synthesized the eugenol based derivatives and investigated them for human MAO inhibitory potential as promising candidates for therapeutics of neurological disorders. Moreover, radical scavenging activity of designed derivatives was tested by and H2O2 and DPPH scavenging methods. Eugenol based derivatives were designed and synthesized for human MAO inhibitory action. The in silico and in vitro models were utilized for the evaluation of hMAO inhibition. The insight into molecular interactions among the compounds and both hMAO-A and hMAO-B active site was achieved by molecular docking studies. The two spectrophotometric titrations techniques were used to evaluate antioxidant potential. Compounds 5b and 16 were found as most active hMAO-A inhibitors with IC50 values of 5.989 ± 0.007 µM and 7.348 ± 0.027 µM respectively, through an appreciable selectivity index value of 0.19 and 0.14 respectively. In case of hMAO-B inhibition compounds 13a and 13b were found as most active hMAO-B inhibitors with IC50 values of 7.494 ± 0.014 µM and 9.183 ± 0.034 µM receptively and outstanding value of selectivity index of 5.14 and 5.72 respectively. Radical scavenging assay showed that compounds 5b, 5a, 9b, 9a were active antioxidants. The findings of present study indicated excellent correlation among dry lab and wet lab hMAO inhibitory experiments. Interestingly, the compounds exhibiting better MAO inhibition activity was also appeared as good antioxidant agents.
Background
Nowadays major depressive disorders are affecting more than 12% population throughout the world in the form of stress and obsessive mood changes [1]. Around half of the all age group is enduring major depressive disorder (MDD) knowingly or unknowingly [2]. Most of the population with MDD generally not adequately responds to regularly approved drugs, for instance selective serotonin reuptake inhibitors (SSRIs). Moreover, millions of people worldwide are affected by neurodegenerative diseases [3]. Parkinson’s disease and Alzheimer’s disease are of the mainly widespread category, and among above five million Americans suffering with Alzheimer’s disease, and as a minimum 400,000 Americans suffering with Parkinson’s disease, though several approximations are much high [4]. Consequently, there is urgent need to develop effective anti-neurodegenerative and antidepressants drug based on natural products due to their lesser side effects. The inhibition of monoamine oxidase (MAO) is a significant way to treat neurodegenerative and depressive disorders.
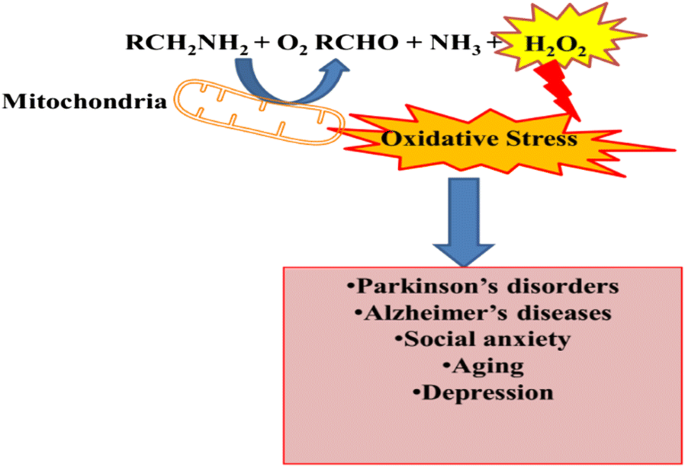
Monoamine oxidase (MAO; EC1.4.3.4) are mitochondrial membranous enzymes that catalyze the oxidative deamination of neurological catecholamines in the peripheral tissues and brain. The targeting of MAO is implicated in numerous mental disorders and renders psychological effects of some neurological drugs [5]. The metabolic by-products of reaction catalyze by MAO generates the corresponding aldehyde and ammonia along with hydrogen peroxide H2O2 which are highly neurotoxic [6] (Fig. 1). The overexpression of MAO or excess action by MAO initiates the neurodegenerative process, anxiety and depression. The resulted aldehyde molecule from deamination is associated with α-synuclein aggregation [7]. That leads to the Parkinson’s disease pathogenesis, however the H2O2 overproduction cause the oxidative damage and aggravates the apoptotic signaling actions. The MAO-A inhibition is associated to antidepressant activity, whereas inhibitors of MAO-B are related to the Parkinson’s disease treatment [8].
Moreover, the presently used MAO inhibitors are associated with numerous adverse effects, as hypertensive crises and liver toxicity [9]. Thus, the development of safe and effective MAO inhibitors is a challenge for the medicinal chemists to serve some potent and selective MAO inhibitors for the treatment of the neuropsychological and neurodegenerative disorders. The development of 3D crystallographic structure by Binda and coworkers has increased the enthusiasm of medicinal chemists to design more effective and targeted leads as MAO inhibitors [10]. By taking the advantage of modern computational methods such as molecular docking, absorption, distribution, metabolism, excretion and toxicity (ADMET), and QSAR along with synthetic medicinal chemistry might provide the target selective and effective MAO inhibitors.
Being the member of phenylpropanoids eugenol is chemically a substituted guaiacol via allyl chain. Eugenol is generally seeming as colorless to light yellow, oily liquid as aromatic essential oils extracted from basil, nutmeg, bay leaf, clove oil and cinnamon and its name is originated from Eugenia caryophyllata [11]. The general biosynthesis of eugenol occurs via amino acid tyrosine through sinapyl-alcohol dehydrogenase (SAD) forming coniferyl acetate and ultimately eugenol synthase to eugenol [12]. It is also worth to state the enormous work has been reported on natural phenols on MAO inhibition along with synthetic modifications and computational studies. Eugenol has attracted considerable attention because of its potential anticonvulsive, neuroprotective, anti-neurodegenerative activities and antidepressant. Moreover, many of these quoted the role of MAO for the neurological effects produced by eugenol [13]. Several reports also indicated the profound anti-oxidative effects of eugenol to reduce oxidative stress which is major cause of neurodegenerative disorders. Kong and coworkers evaluated the four phenols paeonol, honokiol, magnolol and eugenol for the MAO inhibitory potential from mitochondrial rat brain. Among all evaluated natural phenols eugenol showed significant MAO inhibitory potential [14] (Fig. 2).
Moreover, Clarke and coworkers evaluated eugenol from different biological sources as clove, oregano, nutmeg and cinnamon and administered to mice for 14 days. Eugenol significantly increased the forced swim test (FST) score. These observations show that eugenol cross the activity in a dose dependent manner blood–brain barrier in a small amount and act as a competitive human MAO-A inhibitor (Ki = 25 µM) and MAO B (Ki = 211 µM) [15]. Stafford et al. [16] further showed that the eugenol as a major active constituent within Mentha aquatica has remarkably inhibited MAO-B isoform by leaf extract within ethyl acetate at an IC50 value of 6 ± 5 μg/mL and within petroleum ether = 2 ± 1 μg/mL).
In another report by Klein-Júnior et al. [17] essential oils extracted from Eryngium species having eugenol as principle constituent showed notable monoamine oxidase inhibitory activity with IC50 value of 5.65 mg/mL. This study supported the fact that the natural species of Eryngium has eugenol as a chief bioactive secondary metabolite that potentially act on the central nervous system and could be promising drug agent for the treatment of neurodegenerative disorders. A comparative study conducted by Iriea and coworkers showed that eugenol display antidepressant-like activity via tail suspension test and forced swim test (FST) in mice better than imipramine (tricyclic antidepressant). The extracted eugenol from Rhizoma acori graminei stimulated hippocampus brain-derived neurotrophic factor (BDNF) and showed antidepressant-like activity which was not observed as with imipramine [18]. Similarly, Sousa and coworkers demonstrated the eugenol form S. aromaticum (L.) Merr. renders the antidepressant-like actions via inhibiting monoamine neurotransmission (MAO-A) at the lethal dose (LD50 = 4.5 g/kg) in mice. Moreover, repeated administration of eugenol decreased immobility in the FST and tail suspension test (TST) in a dose-dependent mode in mice [19] (Table 1).
Furthermore, some studies have postulated the mechanism of its neurological actions for e.g. Dubey et al. [20], explored the effect of eugenol on the amyloid formation of selected globular proteins. They revealed the neuroprotective nature of eugenol against toxic amyloids by stabilizing native proteins and to delay the conversion of protein species of native conformation into β-sheet assembled mature fibrils, which seems to be crucial for its inhibitory effect. Survey of compounds structurally related to eugenol has identified a few that inhibit MAOs more potently. Together, the activity of eugenol Mechan et al. [21] measured the MAO-A inhibition of oregano extract containing carvacrol. Both of the constituents exhibited selective MAO-A inhibitory activity in a dose dependent manner.
Tao and coworkers evaluated the eugenol from Rhizoma acori graminei for MAO inhibitory action and established the docking poses [22]. Eugenol showed inhibition constant for hMAO-A as Ki = 26 µM and Ki = 15 µM for hMAO-B. Docking simulation studies showed that eugenol interacted mainly with Tyr197, Tyr407, Asn181, Tyr444, Tyr69, and Gly443 active residues within the dynamic site. The aromatic phenolic ring was sandwiched between aromatic cases formed by Tyr444 and Tyr407 and this geometrical position lead to π–π stacking. Moreover, within the MAO-B active site, the aromatic phenolic ring found as embedded within Leu171 and Gln206 side chain.
Inspired by above-mentioned studies and their outcomes we designed and synthesized the eugenol based derivatives as MAO inhibitors (Fig. 3). Moreover, their radical scavenging activity was also evaluated to overcome oxidative stress. The molecular docking studies were performed to rationalize the selectivity for both MAO isoforms.
Results and discussion
Chemistry
The key starting material, 2-(4-allyl-2-methoxyphenoxy)acetohydrazide (3) was prepared according to published procedures by Al-Amiery et al. [23]. The synthesis was initiated from eugenol (1) which is commercially available from Hi-media. The synthesis of ethyl 2-(4-allyl-2-methoxyphenoxy) acetate (2) was carried out by refluxing ethyl chloroacetate with eugenol in the presence of anhydrous K2CO3 in DMF at 80 °C for 24 h (Scheme 1). The confirmation of the formation of ethyl 2-(4-allyl-2-methoxyphenoxy)acetate (2) was evaluated by single spot TLC under UV lamp and IR as the phenolic 3400 cm−1 has been disappeared and the peak at 1735 cm−1 appeared to indicated formation of ester (Table 2). Moreover, the formation of ester was tested by 1HNMR indicating the disappearance of singlet 5.0 of phenolic OH. Finally, the 2-(4-allyl-2-methoxyphenoxy)acetohydrazide (3) was synthesized by refluxing ethyl 2-(4-allyl-2-methoxyphenoxy)acetate (2) with hydrazine hydrate in the presence of ethanol at 80 °C for 2–5 h. The IR spectrum of compound (3) confirmed a carbonyl group at 1739 cm−1, along with hydrazine amide group at 3268 cm−1. In the 1H-NMR; of compound (3) the allyl C–H (alkene) protons appeared at 6.43 ppm (singlet) and 5.83 ppm (singlet) and the singlet (NH) at 5.24 ppm.
Finally, 5(a–b) and compound 7 were prepared with compound (3) and different acid chlorides in the presence of ammonium thiocyanide by refluxing at 100 °C for 4–6 h (Table 3). The reaction completion was confirmed by TLC using the solvent systems methanol-ethyl acetate–benzene (30:30:40 v/v/v). And the single spots were visualized under UV lamp (255 and 360 nm). The 1H-NMR spectrum exhibited a doublet at 8.72–7.95 indicating the presence of –CO–NH–NH–SO– proton for e.g. compound 5a. For compound 7, the IR spectrum has the following characteristic absorption bands: 3377 (C=H str., Ar), 1614 (C=O str., 20amide), 1177 (C–O–C assym. str., Ar), 1027 (C=S str., thiosemicarbazide), 945 (N–N str., N–NH). The 13CNMR peak at 178.9 indicated the presence of C=S bond formation in compound 7. The 1H-NMR spectrum exhibited 7.65–7.59 multiplet showed the presence of secondary amide and at 3.51 doublet specified the amine group formation.
The compounds 11 and 9(a–b) were synthesized by (3) in the presence of nicotinoyl chloride and other natural based acid chlorides via reflux at 80 °C for 8 h in acetone. The IR peaks at 3440 indicated: phenolic O–H, 2960 cm−1 aromatic C=H, 1688 cm−1 secondary amide C=O, 1205 cm−1 aromatic C–O–C group, 1064 cm−1 thiosemicarbazide C=S, 917 cm−1 N–NH as N–N in compound 9a. In the 1H-NMR spectrum doublet peaks at around 7.70 indicated the two secondary amide groups in compound 9a. In case of compound 11 the IR peaks found as 3388 cm−1 for aromatic C=H bond, 1636 cm−1 N=CH for C=N group, 1614 cm−1 for secondary amide C=O, 1229 cm−1 for aromatic C–O–C group, 913 cm−1 N–NH for N–N groups. The ester derivatives 16, 17, 13(a–d) were synthesized by refluxing eugenol with different natural based acid chlorides in the presence of pyridine at 80–90 °C for 8 h in diethyl ether. The confirmation for etherification reaction was done by single spot TLC using the solvent systems acetone–toluene (30:70, v/v). The IR spectra for compound 11 was found as 3077 cm−1 indicating aromatic C=H, 1700 cm−1 as ester formation as carbonyl group and 1206 cm−1 shown the aromatic C–O–C group. Moreover, the elemental analysis and mass spectrometric data were also found in agreement with compounds characterization.
In vitro human MAO-A and human MAO-B inhibition
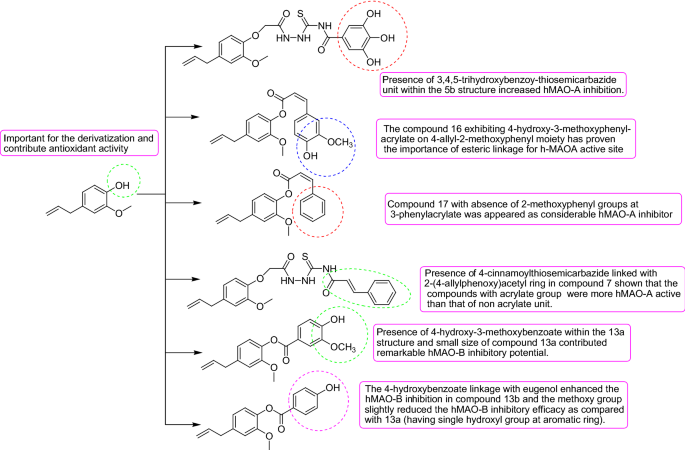
The results of the enzymatic experiments of the eugenol based derivatives compounds 5b and 16 were found as most active hMAO-A inhibitors with IC50 values of 5.989 ± 0.007 µM and 7.348 ± 0.027 µM respectively, through an appreciable selectivity index value of 0.19 and 0.14 respectively (Table 4). 3,4,5-trihydroxybenzoy-thiosemicarbazide unit within the 5b structure. Along with the location of carboxylic moiety in the 2-(4-allylphenoxy) acetyl nucleus the role of semicarbazides towards MAO inhibition has been observed. Moreover, the compound 16 exhibiting 4-hydroxy-3-methoxyphenyl-acrylate on 4-allyl-2-methoxyphenyl moiety has proven the importance of esteric linkage for h-MAOA active site. However, compound 17 with the absence of 2-methoxyphenyl groups at 3-phenylacrylate has appeared as a considerable hMAO-A inhibitor with an IC50 value 13.92 ± 0.051 µM and selectivity index of 0.40. Compound 7 showed significant hMAO-A inhibition with IC50 value 10.46 ± 0.072 µM and a selectivity index of 0.24. Presence of 4-cinnamoylthiosemicarbazide linked with 2-(4-allylphenoxy)acetyl ring in compound 7 shown that the compounds with acrylate group were more hMAO-A active than that of non acrylate unit. The reference compound chlorgyline showed hMAO-A inhibition with IC50 value 18.74 ± 0.096 µM. It has been reported that the active site of MAO-A is highly hydrophobic in nature which facilitates the large sized structures within it feasibly. So there has been a rational point for design the specific hMAO-A inhibitors with large aromatic structures.
In the case of hMAO-B inhibition compounds 13a and 13b were found as most active hMAO-B inhibitors with IC50 values of 7.494 ± 0.014 µM and 9.183 ± 0.034 µM receptively and outstanding value of selectivity index of 5.14 and 5.72 respectively. Presence of 4-hydroxy-3-methoxybenzoate within the 13a structure and small size of compound 13a contributed considerable hMAO-B inhibitory potential. In the case of compound 13b the 4-hydroxybenzoate linkage with eugenol enhanced the hMAO-B inhibition. It was observed that methoxy group slightly reduced the hMAO-B inhibitory efficacy as compared with 13a (having a single hydroxyl group at the aromatic ring). Moreover, the reference compound pargyline exhibited the value of 7.494 ± 0.014 µM for hMAO-B inhibition. As it is documented that hMAO-B enzyme composes of two cavities one is entrance cavity whereas another is substrate cavity, the amino acid residue Ile199 act as “gate keeper” for the substrate/inhibitor, so it depends upon the conformation of Ile199 (closed or open) for the interaction of the inhibitor compound. Moreover, the compounds 13a and 13b are smaller than other structures so they might be easily assessed the entrance cavity for inhibition.
Enzyme kinetic for MAO inhibition
On the basis of the finding that compounds 5b and 13a are most potent hMAO-A and hMAO-B inhibitors, Lineweaver–Burk plots were constructed to understand the mechanism of inhibition MAO-A and MAO-B by these compounds. The Michaelis–Menten kinetics for both isozymes at their predetermined initial rates of reaction is presented as Lineweaver–Burk plot. Meanwhile, For each inhibitor, a set of six plots was constructed by employing eight different concentrations (15–250 μM) of the substrate (p-tyramine) for each plot. The inhibitor concentrations selected for the six plots were built. The intersection of all linear regression lines at y-axis and not x-axis indicates competitive mode of inhibition. These Lineweaver–Burk plots are shown in Figs. 4 and 5, and were indicative of a competitive inhibition of MAO-A by both compounds 5b and 13a.
Statistical analysis
The outcomes of all studies are examined as mean ± SEM (standard error mean). Investigational results were calculated statistically by applying the analysis of one-way variance (ANOVA). Though, ANOVA showed significant variation as computed by ANOVA/Dunnett’s test. The p < 0.05 was observed as to be statistically considerable. The valuation of statistical parameters was performed by Graph Pad Prism 5.0 edition designed for Windows (San Diego, CA, USA).
Docking studies
Molecular docking study was performed to insight the enzyme-inhibitor interactions for structural requirements for MAO activity and selectivity of eugenol based derivatives. Most active compound 5b exhibited backbone hydrogen bonds Val93 via phenolic OH of 3,4,5-trihydroxybenzoyl unit. Moreover, one edge to face π–π stacking interaction (inter planer distance of ~ 5.04 Å) of 4-allyl-2-methoxyphenoxy and Phe112 was also appeared to support the ligand within the hMAO-A active site. The docking pose of compound 16 within the hMAO-A active site revealed a side chain hydrogen bond interaction with polar amino acid residue Asn181 through the hydroxyl group of the 4-hydroxy-3-methoxyphenyl-acrylate unit at inter planer distance of ~ 2.71 Å. Formation of an edge to face π–π stacking interaction between Phe108 and eugenol aromatic ring supported the ligand within the substrate cavity by inter planer distance of ~ 5.05 Å. Moreover, the ligand 16 was found as embedded within the active site through hydrophobic residues Cys323, Ala111, Val210, Phe108, Leu97, Ile180, and Tyr197 via eugenol aromatic ring. Some polar residues such as Gln215, Ser209, and Thr336 were found as engaged towards ester linkage of 16. The 4-hydroxy-3-methoxyphenyl-acrylate unit was surrounded by an aromatic ring formed by Tyr407 and Tyr444, though the hydrophobic residues Leu337, Ile35, Tyr69, Phe352, and Tyr197 were also appeared near to compound 16. Another hMAO-A active compound 9a formed backbone hydrogen bonding with Val210 through oxygen of eugenol aromatic ring with inter planer distance of ~ 1.76 Å. The NH group of 2-hydroxybenzohydrazide formed another hydrogen bond with Phe208 via inter planer distance of ~ 2.23 Å. Moreover, the 2-hydroxybenzohydrazide unit of compound 9a was supported by hydrophobic residues through Tyr407, Phe352, Met350, Ile180, Ile325, Leu337, Phe108, Cys323 and Ile335. Polar residues such as Gln215, Ser209, Asn212, Thr205, and Thr336 were attracted towards hydrazide linkage of compound 9a.
Docking pose of compound 7 showed an edge to face π–π stacking interaction between Phe108 and eugenol aromatic ring with inter-planer distance of ~ 5.34 Å. Interestingly, the eugenol unit was ideally accommodated within the active site by hydrophobic residues Cys323, Ile335, Met350, Phe352, Tyr69, Tyr407, Ile180, Ala111, Leu97 (Fig. 6).
In case of hMAO-B docking insight of compounds 13a and 13b showed two edges to face π–π stacking interaction between Tyr326 and both aromatic rings of compounds 13a and 13b through inter planer distance of ~ 5.24 Å and ~ 5.36 Å for 13a and ~ 5.29 Å and ~ 5.37 Å for 13b respectively. This outstanding behavior of 13a and 13b was noticed due to the small size of the compounds 13a and 13b as it has been reported that the active site of hMAO-B composes of two bipartite cavities. The inhibitor/substrate has to cross through two hydrophobic sub-cavities; the entrance cavity through dimensions of ~ 290 Å as well as the substrate cavity through dimensions of ~ 400 Å. Moreover both compounds 13a and 13b were surrounded by hydrophobic residues such as Phe168, Trp119, Leu167, Phe103, Pro104, Leu88, Pro102, Cys172, and Leu171. The aromatic case formed by residues Tyr188, Tyr398, Tyr435, Phe343; Tyr60 enclosed the 4-hydroxy-3-methoxybenzoate and 4-hydroxybenzoate units of 13a and 13b respectively. Most of the compound including eugenol showed hydrogen bonding with Pro102, and some of them interacted with gate keeper residue Ile199. The role of this gate keeper residue cannot be excluded from the discussion. The expedition of a substrate or inhibitor begin in the course of bendable loop (residue 99–112) which works as a gating residue at the outer mitochondrial membrane surface; the side chain isoleucine (Ile199) could exhibit two diverse conformations, closed conformation (separate both cavities) or open conformation (fuse both cavities) based on the character of the docked ligand. Maximum ligands showed edges to face π–π stacking with Trp119 and Tyr326.
In silico ADMET profile of eugenol derivatives
The hypothetical evaluation of adsorption, distribution, metabolism, excretion and toxicity (ADMET) descriptors prediction was carried out by quick prop graphical user interface of Mastro-11.1 Schrödinger (Table 5) [24]. Evaluation of lipophilicity (logP) indicated the octanol–water partition coefficient that must be within limit 5. It could be predicted from the table that all of the eugenol based derivatives shown logP values within the limit. Lesser blood–brain barrier (BBB) access and poor membrane permeability is associated with a larger rate of tPSA (> 145 Å2) [32]. Nearly all of the synthesized derivatives exhibited acceptable values of tPSA within a range of 35–140 (Table 4). Based on Lipinski’s guide of 5 [25], below 5 hydrogen bond donors must be in lead, and the number of hydrogen bond acceptors (HBA) must be under 10. The aforesaid standard meets as well, because except 5b (with 6 HBD) complete series of derivatives exhibited 0–4 HBD and 1–9 HBA. But this criterion is not fully reliable and has been challenged in case of natural potent well known therapeutic agents. All of the derivatives could be considered to exhibits fine oral bioavailability, examined as confirmed by Veber rule [26]. This states that the rotatable bonds magnitude should be correspondent to 10 and tPSA ≤ 140 Å, or else the whole no. of hydrogen bond donor and acceptor (HBA + HBD) must be ≤ 12. It is considerable that not any of derivatives was discarded, and each one of the derivatives proved promising ADMET rank (Table 4).
Determination of DPPH scavenging activity
Results of DPPH scavenging assay showed that compounds 5b, 5a, 9b, 9a were most active antioxidants with IC50 values of 8.304 ± 0.073 µM, 9.152 ± 0.063 µM, 9.293 ± 0.028 µM, 9.514 ± 0.017 µM respectively (Table 6). It was observed that these compounds were possessing a large number of hydrogen donors as NH and OH groups within their structures. Presence of these groups generally considered to be contributors for potential antioxidant effect by polyphenolic compounds. However, the eugenol and reference compound l-ascorbic acid exhibited the IC50 value of 10.29 ± 0.011 and 8.58 ± 0.009 respectively. Other compounds such as compounds 7 and 11 were also appeared to be considerable antioxidants with IC50 values of 9.786 ± 0.012 µM and 11.21 ± 0.044 µM respectively (Fig. 7). It is notable that the compound 7 was observed as a poor agent for DPPH scavenging, the reason for this might be the absence of any OH or NH group with the compound 7 structure.
H2O2 scavenging activity
The H2O2 scavenging assay was carried out for further evaluate the antioxidant potential of synthesized compounds. In cases of H2O2 scavenging the compounds 5b, 5a, 9b, 9a were most active H2O2 scavengers with IC50 values of 8.904 ± 0.034 µM, 9.156 ± 0.002 µM, 9.393 ± 0.017 µM, 9.547 ± 0.051 µM respectively (Table 7). Surprisingly, the compounds with good DPPH scavenging profile were observed as considerable H2O2 scavengers. Thus, the presence of the OH and NH groups is essential for the antioxidant activity. However, eugenol and reference compound l-ascorbic acid exhibited the IC50 values of 9.980 ± 0.012 µM and 8.121 ± 0.082 µM respectively (Fig. 8). Compounds 7 and 11 were showing similar behavior towards H2O2 and the IC50 values of them were 9.755 ± 0.021 µM and 10.33 ± 0.022 µM respectively.
Nitric oxide scavenging activity
To validate the results of above mentioned antioxidant activity assays we carried out the nitric oxide scavenging activity of the titled compounds. Results of nitric oxide scavenging activity showed that the compound 5b was found as most active NO· scavenger with IC50 value of 6.456 ± 0.023 µM as compared with eugenol (IC50 value 11.724 ± 0.003 µM) and reference compound l-ascorbic acid with IC50 values of 7.523 ± 0.005 µM (Table 8). Moreover compounds 5a, 9b, 9a were also observed as mild active NO· scavengers with IC50 values of 7.156 ± 0.002 µM, 8.095 ± 0.018 µM, 9.007 ± 0.014 µM respectively (Fig. 9). The result of the nitric oxide scavenging activity showed similar routine for the antioxidant action as observed with DPPH and H2O2 scavenging profile.
Ferric reducing antioxidant power (FRAP) assay
Assay of ferric reducing antioxidant power (FRAP) assay showed that compound was compound 5b found as most active antioxidant with IC50 value of 5.112 ± 0.009 µM as compared with eugenol (IC50 value 10.627 ± 0.082 µM) and reference compound l-ascorbic acid with IC50 values of 8.112 ± 0.091 µM (Table 9). However compounds 5a, 9a, 9b were also observed as considerable antioxidant agents with IC50 value of 5.934 ± 0.053 µM, 7.234 ± 0.001 µM, 9.912 ± 0.005 µM respectively (Fig. 10).
Structure–activity relationship (SAR) studies
The structure–activity relationship of the evaluated eugenol based derivatives by means of their antioxidant activity and MAO inhibitory activity and results is summarized in Fig. 11.
Experimental
Materials and methods
Unless otherwise noted, the analytical grade chemicals required for synthesis and antioxidant activity procured from Hi-media Laboratories. The biological evaluation of the test drugs on hMAO activity was investigated by measuring their effects on the production of hydrogen peroxide (H2O2) from p-tyramine (a common substrate for hMAO-A and hMAO-B), using the Amplex Red MAO assay kit (Sigma USA) and microsomal MAO isoforms prepared from insect cells (BTI-TN-5B14) infected with recombinant baculovirus containing cDNA inserts for hMAO-A or hMAO-B (Sigma-Aldrich USA) were used as source for the two microsomal MAO isoforms. Reactions were monitored by thin layer chromatography TLC performed on 0.25 mm precoated silica gel plates procured from Merck, and the spots were visualized in iodine chamber and UV, in mobile media TLC-(5a–11)-methanol-ethyl acetate–benzene (30:30:40 v/v/v), (13a–17)-acetone–toluene (30:70, v/v). Melting points were determined in open capillary tubes on a Sonar melting point apparatus. The nuclear magnetic resonance (NMR) spectra 1H NMR and 13C NMR spectra were recorded in DMSO and deuterated CDCl3 respectively on spectrometer (Model: Bruker Avance II 400 NMR) at a frequency of 400 MHz downfield to tetramethylsilane standard and chemical shifts were recorded as d (parts per million) and s, d, t, q, m, dd, td, dt and tt are designated as singlet, doublet, triplet, quartet, multiplet, doublet of doublets, triplet of doublets, doublet of triplets and triplet of triplets respectively. Coupling constants (J) were reported in hertz (Hz). Infrared (IR) spectra were recorded on Perkin Elmer FTIR spectrophotometer by using KBr pellets technique.
General procedure for the synthesis of acid chlorides
A mixture of different aromatic carboxylic acids and thionyl chloride (10 mL) was stirred and refluxed 80 °C for 2–4 h in the presence of pyridine (1–2 drops) till the completion of reaction. Distillation of the reaction mixture removed the excess of thionyl chloride. Finally the purity of resulting product was appropriate to use it directly for the following synthesis.
General procedure for preparation of eugenol esters (13a–17)
The target esters derivatives derived from eugenol, which were prepared by gently refluxing a solution of eugenol in ether (50 mL) with a solution of different aromatic acid chlorides (0.05 mol) in diethyl ether (50 mL).
The reaction mixture was heated on a water bath (80 °C) until the complete removal of hydrogen chloride and completion of reaction was checked by single spot TLC. The shiny white precipitates so obtained were filtered using suction and rendered free from chloride by repeated washing with water and finally purified by recrystallization with acetone.
General procedure for preparation of 2-(4-allyl-2-methoxyphenoxy)acetohydrazide
Synthesis of 2-(4-allyl-2-methoxyphenoxy)acetohydrazide (0.01 mol) was brought about by refluxing (80 °C) ethyl bromoacetate (0.01 mol) with eugenol (0.01 mol) in the presence of anhydrous K2CO3 in dry DMF for 24 h. Which yielded compound ethyl 2-(4-allyl-2-methoxyphenoxy)acetate, further reaction was followed by the addition of hydrazine hydrate (0.014 mol) to yield 2-(4-allyl-2-methoxyphenoxy)acetohydrazide by refluxing 2–5 h at 80 °C. The crude solid was filtered, dried and recrystallised with mixture of chloroform–methanol (1:2).
General procedure for preparation of 1-(2-(4-allylphenoxy)acetyl)-4phenyl-thiosemicarbazides
5(a–b) and compound 7 were synthesized by refluxing (80 °C) the 2-(4-allyl-2-methoxyphenoxy)acetohydrazide (0.01 mol) with the different aromatic acid chlorides (0.01 mol) and cinnamyl chloride (0.01 mol) in the presence of ammonium thiocyanate and acetone for 8 h. Following recrystallisation from ethanol, the target 1-(2-(4-allylphenoxy)acetyl)-4phenyl-thiosemicarbazides were obtained in appreciable yields.
General procedure for preparation of N′-(2-(4-allyl-2-methoxyphenoxy)acetyl)benzohydrazides and N′-(2-(4-allyl-2-methoxyphenoxy)acetyl)picolinohydrazide
Target compound 2-(4-allyl-2-methoxyphenoxy)acetohydrazides (0.01 mol) were prepared in relatively high yields by reacting the appropriately different aromatic carboxylic acids (0.01 mol) and nicotinoyl chloride (0.01 mol) on reflux (80 °C) for 8 h in acetone. After reaction completion, the solid product was washed with ether and recrystallization was done with ethanol to yield corresponding N′-(2-(4-allyl-2 methoxyphenoxy)acetyl)benzohydrazides and N′-(2-(4-allyl-2 methoxyphenoxy)acetyl)picolinohydrazide.
Spectral data
5a 1-(2-(4-allylphenoxy)acetyl)-4-(4-hydroxybenzoyl)thiosemicarbazide IR (KBr pellets) cm−1 3359 (O–H str., phenol), 3070 (C=H str., Ar), 1663 (C=N str., N=CH), 1602 (C=O str., 20 amide), 1273 (C–O–C assym. str., Ar), 1174 (C=S str., thiosemicarbazide), 943 (N–N str., N–NH). 1H NMR (400 MHz, DMSO-d6): 8.72–7.95 (m, 2H), 7.05 (ddd, J = 7.5, 2.2, 1.1 Hz, 4H), 6.99–6.82 (m, 2H), 5.78 (ddt, J = 16.8, 9.9, 6.2 Hz, 1H), 5.19–5.08 (m, 2H), 4.68 (s, 2H), 3.48 (dp, J = 6.2, 1.0 Hz, 2H). 13C NMR (400 MHz, CDCl3): 180.5, 165.6, 163.7, 159.8, 155.2, 136.9, 135.1, 127.9, 127.1, 126.8, 113.9, 113.4 (d, J = 8.8 Hz), 66.9, 45.6; m/z 415.09 [M+1]+; Anal. For C20H21N3O5S: calcd: C, 57.82; H, 5.09; N, 10.11, found: C, 57.80; H, 5.07; N, 10.10.
5b 1-(2-(4-allylphenoxy)acetyl)-4-(3,4,5-trihydroxybenzoyl)thiosemicarbazide IR (KBr pellets) cm−1 3407 (O–H str., phenol), 3286 (C=H str., Ar), 1705 (C=N str., N=CH), 1626 (C=O str., 20 amide), 1177 (C–O–C assym. str., Ar), 1030 (C=S str., thiosemicarbazide), 944 (N–N str., N–NH). 1H NMR (400 MHz, DMSO-d6): 7.11 (d, J = 7.2 Hz, 4H), 6.80–6.72 (m, 2H), 5.99 (tt, J = 13.4, 6.2 Hz, 1H), 5.31 (dddt, J = 21.1, 13.4, 2.1, 1.0 Hz, 2H), 4.68 (s, 2H), 3.45 (dp, J = 6.2, 1.0 Hz, 2H). 13C NMR (400 MHz, CDCl3): 191.5, 174.6, 175.8, 159.2, 147.6, 138.7, 137.9, 134.1, 129.1, 128.3, 115.4 (d, J = 8.8 Hz), 104.7, 65.9, 39.6; m/z 447.12 [M+1]+; Anal. For C20H21N3O7S: calcd: C, 53.68; H, 4.73; N, 9.39, found: C, 53.67; H, 4.72; N, 9.36.
7 (E)-1-(2-(4-allylphenoxy)acetyl)-4-cinnamoylthiosemicarbazide IR (KBr pellets) cm−1 3377 (C=H str., Ar), 1614 (C=O str., 20 amide), 1177 (C–O–C assym. str., Ar), 1027 (C = S str., thiosemicarbazide), 945 (N–N str., N–NH). 1H NMR (400 MHz, DMSO-d6): 7.65–7.59 (m, 4H), 7.52 (dt, J = 15.1, 0.6 Hz, 2H), 7.49–7.37 (m, 4H), 7.33–7.31 (m, 2H), 7.07–6.88 (m, 4H), 6.86–6.80 (m, 4H), 6.76 (d, J = 15.1 Hz, 2H), 5.94 (ddt, J = 16.8, 9.9, 6.2 Hz, 2H), 5.18–5.11 (m, 3H), 5.07 (dt, J = 2.1, 1.0 Hz, 1H), 4.56 (s, 4H), 3.51 (dp, J = 6.2, 1.0 Hz, 4H). 13C NMR (400 MHz, CDCl3): 178.9, 169.6, 163.8, 157.2, 140.9, 138.9, 135.0 (d, J = 17.5 Hz), 131.1, 129.0 (d, J = 18.3 Hz), 127.1, 122.3, 117.4 (d, J = 8.8 Hz), 70.9, 39.6; m/z 447.12 [M+1]+; Anal. For C22H23N3O4S: calcd: C, 62.10; H, 5.45; N, 9.88, found: C, 62.09; H, 5.44; N, 9.87.
9a N′-(2-(4-allyl-2-methoxyphenoxy)acetyl)-2-hydroxybenzohydrazide IR (KBr pellets) cm−1 3440 (O–H str., phenol), 2960 (C=H str., Ar), 1688 (C=O str., 20 amide), 1205 (C–O–C assym. str., Ar), 1064 (C=S str., thiosemicarbazide), 917 (N–N str., N–NH). 1H NMR (400 MHz, DMSO-d6): 7.70 (dd, J=7.4, 1.5 Hz, 2H), 7.43 (td, J = 7.5, 1.5 Hz, 2H), 7.01–6.93 (m, 4H), 6.88 (dt, J = 7.7, 0.8 Hz, 2H), 6.85 (dp, J = 7.8, 1.0 Hz, 4H), 5.55 (dd, J = 16.8, 9.9, Hz, 2H), 5.15–5.09 (m, 3H), 5.03 (dt, J = 2.2, 1.0 Hz, 1H), 4.77 (s, 4H), 3.38 (s, 6H), 3.31 (dp, J = 6.2, 1.0 Hz, 4H). 13C NMR (400 MHz, CDCl3): 170.7, 167.7, 157.7, 152.8, 151.1, 145.2, 144.1, 143.2, 129.4, 124.2, 117.4, 115.5 (d, J = 5.5 Hz), 113.5, 113.2, 111.3, 67.8, 57.7, 41.4; m/z 356.12 [M+1]+; Anal. For C19H20N2O5: calcd: C, 64.04; H, 5.66; N, 7.86, found: C, 64.03; H, 5.65; N, 7.85.
9b N′-(2-(4-allyl-2-methoxyphenoxy)acetyl)-2-mercaptobenzohydrazide IR (KBr pellets) cm−1 3378 (O–H str., phenol), 3094 (C=H str., Ar), 1716 (C=O str., 20 amide), 1260 (C–O–C assym. str., Ar), 917 (N–N str., N–NH), 697 (C=S str., thiol). 1H NMR (400 MHz, DMSO-d6): 7.72 (dd, J = 7.4, 1.5 Hz, 2H), 7.48 (dd, J = 7.4, 1.6 Hz, 2H), 7.38 (td, J = 7.5, 1.5 Hz, 2H), 7.25 (td, J = 7.4, 1.6 Hz, 2H), 6.88 (dt, J = 7.7, 0.8 Hz, 2H), 6.75 (dp, J = 7.8, 1.0 Hz, 4H), 5.96 (ddt, J = 16.8, 9.9, 6.2 Hz, 2H), 5.15–5.13 (m, 3H), 5.10 (dt, J = 2.2, 1.0 Hz, 1H), 4.73 (s, 4H), 3.87 (s, 6H), 3.35 (dp, J = 6.2, 1.0 Hz, 4H). 13C NMR (400 MHz, CDCl3): 169.7, 165.2, 150.8, 146.1, 139.2, 137.1, 135.8, 132.5, 130.8, 128.4 (d, J = 16.0 Hz), 127.5, 120.2, 115.3, 113.5, 110.3, 67.8, 55.7, 40.4; m/z 372.10 [M+1]+; Anal. For C19H20N2O4S: calcd: C, 61.27; H, 5.41; N, 7.52, found: C, 61.25; H, 5.40; N, 7.51.
11 N′-(2-(4-allyl-2-methoxyphenoxy)acetyl)picolinohydrazide IR (KBr pellets) cm−1 3388 (C = H str., Ar), 1636 (C=N str., N=CH), 1614 (C=O str., 20 amide), 1229 (C–O–C assym. str., Ar), 913 (N–N str., N–NH). 1H NMR (400 MHz, DMSO-d6): 8.76–8.69 (m, 2H), 8.10–8.00 (m, 4H), 7.81 (ddd, J = 7.5, 5.9, 3.1 Hz, 2H), 6.89 (dt, J = 7.7, 0.9 Hz, 2H), 6.77 (dp, J = 7.8, 1.0 Hz, 4H), 5.99 (ddt, J = 16.8, 9.9, 6.2 Hz, 2H), 5.18–5.10 (m, 3H), 5.09 (dt, J = 2.2, 1.0 Hz, 1H), 4.79 (s, 4H), 3.85 (s, 6H), 3.30 (dp, J = 6.2, 1.0 Hz, 4H). 13C NMR (400 MHz, CDCl3): 170.7, 166.7, 151.6, 150.8, 150.6, 146.1, 141.2, 140.9, 140.1, 125.1, 123.1 (d, J = 16.0 Hz), 117.3, 115.5, 112.3, 79.8, 55.7, 40.4; m/z 341.11 [M+1]+; Anal. For C18H19N3O4: calcd: C, 63.33; H, 5.61; N, 12.31, found: C, 63.30; H, 5.59; N, 12.30.
13a 4-allyl-2-methoxyphenyl 4-hydroxy-3-methoxybenzoate 3430 (O–H str., phenol), 3078 (C=H str., Ar), 1733 (C=O str., ester), 1233 (C–O–C assym. str., Ar). 1H NMR (400 MHz, DMSO-d6): 7.69 (dd, J = 7.5, 1.5 Hz, 2H), 7.54 (d, J = 1.5 Hz, 2H), 7.11 (d, J = 7.4 Hz, 2H), 6.84–6.79 (m, 6H), 5.98 (ddt, J = 16.8, 9.9, 6.2 Hz, 2H), 5.19–5.14 (m, 3H), 5.10 (dt, J = 2.2, 1.0 Hz, 1H), 3.84 (d, J = 10.6 Hz, 12H), 3.34 (dp, J = 6.2, 1.0 Hz, 4H). 13C NMR (400 MHz, CDCl3): 163.8, 151.9, 151.8, 144.6, 141.1, 139.8, 138.2, 123.5, 121.5 (d, J = 5.5 Hz), 120.4, 119.9, 116.3, 112.1 (d, J = 13.9 Hz), 56.1, 40.7; m/z 314.09 [M+1]+; Anal. For C18H18O5: calcd: C, 68.78; H, 5.77 found: C, 68.77; H, 5.75.
13b 4-allyl-2-methoxyphenyl 4-hydroxybenzoate IR (KBr pellets) cm−1 3450 (O–H str., phenol), 3077 (C=H str., Ar), 1708 (C=O str., ester), 1245 (C–O–C assym. str., Ar). 1H NMR (400 MHz, DMSO-d6): 7.94–7.82 (m, 2H), 7.12 (d, J = 7.5 Hz, 1H), 6.95–6.92 (m, 2H), 6.83–6.79 (m, 2H), 5.87 (tt, J = 13.4, 6.2 Hz, 1H), 5.14 (dddt, J = 21.2, 13.4, 2.1, 1.0 Hz, 2H), 3.80 (s, 3H), 3.35 (dp, J = 6.2, 1.0 Hz, 2H). 13C NMR (400 MHz, CDCl3): 161.7 (d, J = 3.2 Hz), 149.9, 140.1, 139.8, 137.2, 132.6, 120.6–120.2 (m), 117.4, 117.3, 114.6, 55.9, 40.4; m/z 284.09 [M+1]+; Anal. For C17H16O4: calcd: C, 71.82; H, 5.67 found: C, 71.80; H, 5.65.
13c 4-allyl-2-methoxyphenyl 2-hydroxybenzoate IR (KBr pellets) cm−1 3444 (O–H str., phenol), 3076 (C=H str., Ar), 1747 (C=O str., ester), 1252 (C–O–C assym. str., Ar); 1H NMR (400 MHz, DMSO-d6): 7.99 (dd, J = 7.5, 1.5 Hz, 2H), 7.62 (td, J = 7.5, 1.5 Hz, 2H), 7.12 (d, J = 7.5 Hz, 2H), 7.09 (td, J = 7.5, 1.5 Hz, 2H), 6.91 (dd, J = 7.5, 1.5 Hz, 2H), 6.85–6.77 (m, 4H), 5.99 (ddt, J = 16.8, 9.9, 6.2 Hz, 2H), 5.19–5.11 (m, 3H), 5.07 (dt, J = 2.2, 1.0 Hz, 1H), 3.87 (s, 6H), 3.22 (dp, J = 6.2, 1.0 Hz, 4H). 13C NMR (400 MHz, CDCl3): 170.6, 160.5, 152.9, 139.8, 139.5, 138.2, 135.7, 131.6, 119.5 (d, J = 5.5 Hz), 119.8, 118.5, 115.3, 114.8, 113.6, 56.9, 41.4; m/z 284.1 [M+1]+; Anal. For C17H16O4: calcd: C, 71.82; H, 5.67 found: C, 71.81; H, 5.65.
13d 4-allyl-2-methoxyphenyl 2-mercaptobenzoate IR (KBr pellets) cm−1 3344 (C=H str., Ar), 1717 (C=O str., ester), 1260 (C–O–C assym. str., Ar), 643 (C=S str., thiol). 1H NMR (400 MHz, DMSO-d6): 8.79 (dd, J = 7.4, 1.6 Hz, 2H), 7.56 (dd, J = 7.4, 1.6 Hz, 2H), 7.47 (td, J = 7.4, 1.6 Hz, 2H), 7.34 (td, J = 7.4, 1.6 Hz, 2H), 7.12 (d, J = 7.5 Hz, 2H), 6.90–6.78 (m, 4H), 5.99 (ddt, J = 16.8, 9.9, 6.2 Hz, 2H), 5.17–5.11 (m, 3H), 5.09 (dt, J = 2.2, 1.0 Hz, 1H), 3.86 (s, 6H), 3.24 (dp, J = 6.2, 1.0 Hz, 4H). 13C NMR (400 MHz, CDCl3): 165.4, 150.9, 140.8, 140.5, 137.2, 136.5, 133.1, 131.5, 130.8, 128.8, 125.4, 121.5 (d, J = 5.5 Hz), 117.3, 112.6, 59.9, 47.5; m/z 300.06 [M+1]+; Anal. For C17H16O3S: calcd: C, 67.98; H, 5.37 found: C, 67.97; H, 5.35.
16 (Z)-4-allyl-2-methoxyphenyl 3-(4-hydroxy-3-methoxyphenyl)acrylate IR (KBr pellets) cm−1 3509 (O–H str., phenol), 3075 (C=H str., Ar), 1720 (C=O str., ester), 1206 (C–O–C assym. str., Ar). 1H NMR (400 MHz, DMSO-d6): 7.55 (d, J = 1.6 Hz, 1H), 7.25–7.17 (m, 1H), 7.09 (d, J = 7.8 Hz, 1H), 6.99 (ddd, J = 7.5, 1.5, 0.6 Hz, 1H), 6.76 (tdd, J = 3.5, 1.5, 0.8 Hz, 2H), 6.72 (d, J = 7.5 Hz, 1H), 6.18 (d, J = 10.9 Hz, 1H), 5.94 (ddt, J = 16.8, 9.9, 6.2 Hz, 1H), 5.20–5.11 (m, 1H), 5.11–5.06 (m, 1H), 3.85 (d, J = 10.6 Hz, 6H), 3.32 (dp, J = 6.2, 1.0 Hz, 2H). 13C NMR (400 MHz, CDCl3): 164.4, 151.3, 149.6, 148.8, 144.8, 140.2, 139.8, 138.2, 126.2, 123.9, 121.6, 120.8, 116.3, 115.6, 115.1, 113.2, 111.8, 56.2, 55.9, 40.4; m/z 340.11 [M+1]+; Anal. For C20H20O5: calcd: C, 70.57; H, 5.92 found: C, 70.55; H, 5.91.
17 (Z)-4-allyl-2-methoxyphenyl 3-phenylacrylate IR (KBr pellets) cm−1 3077 (C=H str., Ar), 1700 (C=O str., ester), 1206 (C–O–C assym. str., Ar). 1H NMR (400 MHz, DMSO-d6): 7.61 (ddt, J = 6.5, 1.6, 0.7 Hz, 2H), 7.41–7.36 (m, 1H), 7.36–7.27 (m, 2H), 7.18 (dt, J = 10.9, 0.7 Hz, 1H), 7.10 (d, J = 7.8 Hz, 1H), 6.88 (tdd, J = 3.6, 1.5, 0.8 Hz, 2H), 6.13 (d, J = 10.9 Hz, 1H), 5.80 (tt, J = 13.4, 6.2 Hz, 1H), 5.13 (dddt, J = 21.2, 13.4, 2.1, 1.0 Hz, 2H), 3.78 (s, 3H), 3.29 (dp, J = 6.2, 1.0 Hz, 2H). 13C NMR (400 MHz, CDCl3): 159.5, 153.3, 145.2, 142.2, 141.8, 137.2, 134.6, 130.1, 128.8, 128.5, 122.6, 120.8, 116.9 (d, J = 13.3 Hz), 113.2, 55.9, 41.4; m/z 294.11 [M+1]+; Anal. For C19H18O3: calcd: C, 77.53; H, 6.16 found: C, 77.51; H, 6.14.
Recombinant human MAO inhibition screening
To evaluate IC50 values for the inhibition of human MAO-A and MAO-B by newly synthesized compounds the commercially available recombinant human enzymes expressed in insect cells were used (Sigma-Aldrich). The MAO inhibition assay was based on the method described by Anderson et al., 1993 and Chimenti et al. with some modifications [27, 28]. The Amplex Red MAO Kit tyramine (Sigma-Aldrich) affords a suitable fluorimetric method to evaluate MAO enzyme action of synthetic and natural compounds. In the assay, MAO reacts with p-tyramine, a common substrate for both MAO isoforms, ensuing in the production of H2O2, which is identified by a fluorimetric method (λex = 530/λem = 585 nm). Each test well contained 0.1 mL of sodium phosphate buffer (0.05 M, pH 7.4), a range of concentrations of the eugenol based derivatives or reference compounds along with recombinant hMAO-A or hMAO-B in appropriate quantity. After the preparation of this mixture the wells were subjected to incubation for 15 min at 37 °C, placed in the dark fluorimeter chamber. Once this incubation period over, the process was rendered by mixing 200 lM Amplex Red reagent (Sigma-Aldrich), 1 U/mL horseradish peroxidase (Sigma-Aldrich) and p-tyramine (Sigma-Aldrich). Subsequently, the formation of hydrogen peroxide exerted by MAO isoforms was quantified by using Amplex-Red reagent, a non-fluorescent probe that reacts with H2O2 in the existence of horse radish peroxidase to generate the fluorescent component resorufin. Moreover, the control analysis was calculated in the absence of compound and reference inhibitors. The probability of compounds to change the fluorescence produced in the reaction mix due to non-enzymatic inhibition was assessed by mixing titled compounds to solutions of single Amplex Red reagent in a sodium phosphate buffer. Data were processed in Microsoft Excel, to calculate IC50 values.
In silico ADME prediction
The in silico ADMET evaluation of the synthesized compounds was performed by QikProp software by Schrödinger suit. Several descriptors for determination of essential pharmacological properties were calculated for instance Lipinski rule, total polar surface area, MDCK, Caco-2, no. of rotatable bonds, logPoct and logBB. Caco-2 cell and Madin–Darby canine kidney (MDCK) cell models are considered crucially unfailing in vitro model to estimate the oral drug absorption. For a CNS acting drug the logBB is an important descriptor to identify blood–brain barrier (BBB) diffusion and it is estimated as the concentration range of compound concentration in peripheral blood (Cblood) and brain (Cbrain) [29,30,31,32,33]. The ADMET score calculated by QikProp showed the crucial parameters for the drug-like properties of the synthesized compounds.
Enzyme kinetics
Lineweaver–Burk plots for the inhibition of MAO-A and MAO-B were constructed by using p-tyramine at eight different concentrations for each plot (15–250 μM). For each test inhibitor, six Lineweaver–Burk plots were constructed. Plotting of Michaelis–Menten curves was used to estimate inhibition data by global fitting of the to the Michaelis–Menten equation using the Prism 5 software package.
Preparation of ligands
The 3D structures for all the eugenol based derivatives were designed in MDL MOL format and then processed by LigPrep module of Schrödinger. The LigPrep utilized to optimize and assign the accurate protonation pose and torsions to all ligands. Finally the stereochemical structures around 32 was done at pH state 7.0 ± 2.0 via utilizing ionizer. The desalting of ligands and optimization and tautomerization was done by utilizing OPLS 2005 force field.
Preparation of proteins
The 3-D crystallographic protein structures for hMAO-A and hMAO-B at good resolution were taken from the Protein Data Bank with succession codes 2Z5X [34] and 2V5Z [35] respectively. Prior to molecular docking the removal of inhibitors as co-crystallized form, safinamide for 2V5Z and harmine for 2Z5X and structural water molecules was done from hMAO-A and hMAO-B proteins. The explicit active sites were precisely set at 1000 A3 standard grid box located near cofactor FAD’s N5 atom. Consequently, the protonation and charge position were allocated and minimization of energy was done via OPLS2005 force field.
Docking methodology
Molecular docking was carried out by implementing flexible algorithm with XP approach of Glide. The grid box was generated frequently for assortment and formation of centroid of the dynamic site residues and the integral receptor protein was processed via energy minimization at RMSD rate of 0.17 Å. The side chains of B-factor in MAO protein were separated before docking experiments. All of the amino acid residues were refined and energy minimization and optimization of primary side chains. The aforementioned process exposed the correct site of ligand structure and conformation for induced fit docking. Therefore, with the XP Glide mode redocking was performed which afforded dock score for every ligand.
Determination of DPPH scavenging activity
The antioxidant activity of the newly synthesized compound was measured on the basis of the scavenging activity of the stable 1, 1-diphenyl 2-picrylhyorazyl (DPPH) free radical The evaluation of the antioxidant activity was based on free radical scavenging effect of stable DPPH· with spectrophotometric titration [36, 37]. Solutions of synthesized compounds were set in ethanol/water (1:1) having 10 mM to 100 mM at a period of 10 mM. The 0.01% DPPH· stock solution was prepared in ethanol/water (1:1). Samples were prepared as 5 mL DPPH· solution was mixed with 5 mL l-ascorbic acid solution then shaken vigorously and kept in the dark. Corresponding blank sample were prepared and l-ascorbic acid was used as reference standard. All samples were subjected for incubation at 37 °C for an hour in the dark chamber prior to evaluate absorbance. To evaluate the absorption spectra, firstly blank spectra for ethanol/water were observed afterward the spectrophotometric titration was carried out at various concentrations of titled compounds. The absorbance was calculated at 517 nm with UV/Vis Epoch ELISA reader, and radical scavenging activity was calculated as a reduction in absorbance of DPPH·. The actual decrease in absorption induced by the test was compared with the positive controls. The IC50 values were calculated use the dose inhibition curve in linear range by plotting the extract concentration versus the corresponding scavenging effect through following equation [(ADR − APR)/AO] × 100. Where ADR is the absorbance prior to reaction and APR is the absorbance after reaction with DPPH·.
Hydrogen peroxide scavenging (H2O2) assay
Hydrogen peroxide generally turns into water and oxygen and produces highly toxic hydroxyl radical that renders the damage to DNA and peroxidation of lipid. H2O2 scavenging ability of eugenol based derivatives was evaluated by method described by Nakano and colleagues with some alteration [38, 39]. The hydrogen peroxide solution (40 mM) was prepared in phosphate buffer (40 mM pH 7.4) and addition of several ranges of synthesized derivatives (05–80 μg/mL) was done into H2O2 solution (2 mL). The determination of H2O2 concentration was done by UV/Vis Epoch ELISA reader at 230 nm upon incubation of 10 min. Solution of phosphate buffer without H2O2 was noted as blank reading. Antioxidant potential of synthesized compounds is shown in Table 3. The hydrogen peroxide percentage inhibition was estimated through the formula [(AH − AD)/AO] × 100, where AH is the absorbance of the control and AD is the absorbance of compounds/reference taken as 05–80 μg/mL l-ascorbic acid.
Nitric oxide scavenging activity
The generation of nitric oxide (NO·) on biological tissues is mediated by particular nitric oxide synthases enzyme that control the catabolism of the arginine to citrulline via creation of NO through a five electron oxidative reaction. At the physiological pH (7.2) in aqueous solution sodium nitroprusside decompose to produce NO·. Finally, NO· reacts with oxygen at underaerobic conditions and produces stable products (nitrate and nitrite), this amount can be determined by means of Griess reagent. The procedure for the nitric oxide scavenging activity was adopted from reports of Alam et al. [40].
Ferric reducing-antioxidant power (FRAP) assay
The capacity of antioxidants to decrease ferric iron is measured by this method. The reaction of 2,3,5-triphenyl-1,3,4-triaza-2-azoniacyclopenta-1,4-diene chloride (TPTZ) and complex ferric iron yields ferrous form at low pH. The monitoring of the above reduction reaction at 593 nm under ELISA reader reveals the antioxidant potential. This method was adopted from the reports of Benzie et al. [41].
Conclusion
All of the research findings facilitated to conclude the structural specificity rationale for the hMAO-B and hMAO-A of this new series of natural eugenol based inhibitors. The results of the study improved our confidence in our scheme and encouraged us to carry on our study to design more persuasive and selective inhibitors. The considerable hMAO-A inhibitory potential of compounds 5b with IC50 value (5.989 ± 0.007 µM) might be due to presence of 3,4,5-trihydroxybenzoy-thiosemicarbazide unit within the 5b structure. Moreover the compound 16 exhibiting 4-hydroxy-3-methoxyphenyl-acrylate on 4-allyl-2-methoxyphenyl moiety has proven the importance of esteric linkage for h-MAOA active site. In case of compound 13b the 4-hydroxybenzoate linkage with eugenol enhanced the hMAO-B inhibition. I was observed that methoxy group slightly reduced the hMAO-B inhibitory efficacy as compared with 13a (having single hydroxyl group at aromatic ring). Together with significant MAO inhibitory activity of abovementioned derivatives the future trend is directed towards pre-clinical studies. The reduction of physiological antioxidants in the CNS causes excessive oxidative stress by overproduction of MAO in brain and is concerned with neurodegenerative disorders. We synthesized the natural eugenol based compounds which exhibits considerable MAO inhibitory potential along with antioxidant activity that might probably pay for foremost advantages to explore the fundamental neurological pathology.
References
Youdim MB, Edmondson D, Tipton KF (2006) The therapeutic potential of monoamine oxidase inhibitors. Nat Rev Neurosci 7:295
Krishnan V, Nestler EJ (2008) The molecular neurobiology of depression. Nature 455:894–895
Barnham KJ, Masters CL, Bush AI (2004) Neurodegenerative diseases and oxidative stress. Nat Rev Drug Discov 3:205
Werber EA, Rabey JM (2001) The beneficial effect of cholinesterase inhibitors on patients suffering from Parkinson’s disease and dementia. J Neural Transm 108:1319–1325
Dhiman P, Malik N, Khatkar A (2018) 3D-QSAR and in silico studies of natural products and related derivatives as monoamine oxidase inhibitors. Curr Neuropharmacol 16:881–900
Zhou M, Panchuk-Voloshina N (1997) A one-step fluorometric method for the continuous measurement of monoamine oxidase activity. Anal Biochem 253:169–174
Dhiman P, Malik N, Khatkar A, Kulharia M (2017) Antioxidant, xanthine oxidase and monoamine oxidase inhibitory potential of coumarins: a review. Curr Org Chem 21:294–304
Dhiman P, Malik N, Khatkar A (2018) Hybrid caffeic acid derivatives as monoamine oxidases inhibitors: synthesis, radical scavenging activity, molecular docking studies and in silico ADMET analysis. Chem Cent J 12:112
Yamada M, Yasuhara H (2004) Clinical pharmacology of MAO inhibitors: safety and future. Neurotoxicology 25:215–221
Binda C, Newton-Vinson P, Hubálek F, Edmondson DE, Mattevi A (2002) Structure of human monoamine oxidase B, a drug target for the treatment of neurological disorders. Nat Struct Mol Biol 9:22–23
Khalil AA, ur Rahman U, Khan MR, Sahar A, Mehmood T, Khan M (2017) Essential oil eugenol: sources, extraction techniques and nutraceutical perspectives. RSC Adv 7:32669–32681
Ferrer JL, Austin MB, Stewart C Jr, Noel JP (2008) Structure and function of enzymes involved in the biosynthesis of phenylpropanoids. Plant Physiol Biochem 46(3):356–370
Niedzielska E, Smaga I, Gawlik M, Moniczewski A, Stankowicz P, Pera J, Filip M (2016) Oxidative stress in neurodegenerative diseases. Mol Neurobiol 53:4094–4125
Kong LD, Cheng CH, Tan RX (2004) Inhibition of MAO A and B by some plant-derived alkaloids, phenols and anthraquinones. J Ethnopharmacol 91:351–355
Clarke SE, Ramsay RR (2011) Dietary inhibitors of monoamine oxidase A. J Neural Transm 118:1031–1041
Stafford GI, Pedersen PD, Jäger AK, Van Staden J (2007) Monoamine oxidase inhibition by southern African traditional medicinal plants. S Afr J Bot 73:384–390
Klein-Júnior LC, dos Santos Passos C, Tasso de Souza TJ, Gobbi de Bitencourt F, Salton J, de Loreto Bordignon SA, Henriques AT (2016) The monoamine oxidase inhibitory activity of essential oils obtained from Eryngium species and their chemical composition. Pharm Biol 54:1071–1076
Irie Y, Itokazu N, Anjiki N, Ishige A, Watanabe K, Keung WM (2004) Eugenol exhibits antidepressant-like activity in mice and induces expression of metallothionein-III in the hippocampus. Brain Res 1011:243–246
de Sousa DP, Silva RH, Silva EF, Gavioli EC (2017) Essential oils and their constituents: an alternative source for novel antidepressants. Molecules 22:1290
Dubey K, Anand BG, Shekhawat DS, Kar K (2017) Eugenol prevents amyloid formation of proteins and inhibits amyloid-induced hemolysis. Sci Rep 7:40744
Mechan AO, Fowler A, Seifert N, Rieger H, Wöhrle T, Etheve S, Wyss A, Schüler G, Colletto B, Kilpert C, Aston J (2011) Monoamine reuptake inhibition and mood-enhancing potential of a specified oregano extract. Br J Nutr 105:1150–1163
Tao G, Irie Y, Li DJ, Keung WM (2005) Eugenol and its structural analogs inhibit monoamine oxidase A and exhibit antidepressant-like activity. Bioorg Med Chem 13:4777–4788
Al-Amiery AA, Musa AY, Kadhum AA, Mohamad AB (2011) The use of umbelliferone in the synthesis of new heterocyclic compounds. Molecules 16:6833–6843
Maestro, version 11.6, Schrödinger, LLC, New York, NY. 2018
Walters WP, Namchuk M (2003) A guide to drug discovery: designing screens: how to make your hits a hit. Nat Rev Drug Discov 2:259
Veber DF, Johnson SR, Cheng HY, Smith BR, Ward KW, Kopple KD (2002) Molecular properties that influence the oral bioavailability of drug candidates. J Med Chem 45(12):2615–2623
Chimenti F, Secci D, Bolasco A, Chimenti P, Bizzarri B, Granese A, Carradori S, Yánez M, Orallo F, Ortuso F, Alcaro S (2009) Synthesis, molecular modeling, and selective inhibitory activity against human monoamine oxidases of 3-carboxamido-7-substituted coumarins. J Med Chem 52:1935–1942
Anderson MC, Hasan F, McCrodden JM, Tipton KF (1993) Monoamine oxidase inhibitors and the cheese effect. Neurochem Res 18(11):1145–1149
Jeffrey P, Summerfield SG (2007) Challenges for blood–brain barrier (BBB) screening. Xenobiotica 37:1135–1151
Skibba M, Hye Khan M, Kolb LL, Yeboah MM, Falck JR, Amaradhi R, Imig JD (2017) Epoxyeicosatrienoic acid analog decreases renal fibrosis by reducing epithelial-to-mesenchymal transition. Front Pharmacol 8:406
Reddy MD, Blanton AN, Watkins EB (2017) Palladium-catalyzed, N-(2-aminophenyl) acetamide-assisted ortho-arylation of substituted benzamides: application to the synthesis of urolithins B, M6, and M7. J Org Chem 82(10):5080–5095
Ganesh T, Banik A, Dingledine R, Wang W, Amaradhi R (2018) Peripherally restricted, highly potent, selective, aqueous-soluble EP2 antagonist with anti-inflammatory properties. Mol Pharm 15(12):5809–5817
Reddy MD, Fronczek FR, Watkins EB (2016) Rh-catalyzed, regioselective, C–H bond functionalization: access to quinoline-branched amines and dimers. Org Lett 18(21):5620–5623
Son SY, Ma J, Kondou Y, Yoshimura M, Yamashita E, Tsukihara T (2008) Structure of human monoamine oxidase A at 2.2-Å resolution: the control of opening the entry for substrates/inhibitors. Proc Natl Acad Sci 105:5739–5744
Binda C, Wang J, Pisani L, Caccia C, Carotti A, Salvati P, Edmondson DE, Mattevi A (2007) Structures of human monoamine oxidase B complexes with selective noncovalent inhibitors: safinamide and coumarin analogs. J Med Chem 50:5848–5852
Malik N, Dhiman P, Verma PK, Khatkar A (2015) Design, synthesis, and biological evaluation of thiourea and guanidine derivatives of pyrimidine-6-carboxylate. Res Chem Intermed 41:7981–7993
Motati DR, Uredi D, Watkins EB (2018) A general method for the metal-free, regioselective, remote C–H halogenation of 8-substituted quinolines. Chem Sci 9(7):1782–1788
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22(5):867–880
Xu Q, Kulkarni AA, Sajith AM, Hussein D, Brown D, Güner OF, Reddy MD, Watkins EB, Lassegue B, Griendling KK, Bowen JP (2018) Design, synthesis, and biological evaluation of inhibitors of the NADPH oxidase, Nox4. Bioorg Med Chem 26(5):989–998
Alam MN, Bristi NJ, Rafiquzzaman M (2013) Review on in vivo and in vitro methods evaluation of antioxidant activity. Saudi Pharm J 21(2):143–152
Benzie IF, Strain JJ (1999) [2] Ferric reducing/antioxidant power assay: direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Methods Enzymol 299:15–27
Authors’ contributions
PD and AK have designed, synthesized and carried out the anti-hMAO and antioxidant activity and NM, have carried out the docking simulations with in silico ADMET studies. All authors read and approved the final manuscript.
Acknowledgements
The authors are extremely grateful to the Head, Department of Pharmaceutical Sciences, M. D. University, Rohtak for providing fundamental facilities to carry out this investigation study. The authors are also thankful to Dr. Vinod Devaraji Application Scientist Schrödinger LLC for his sustain guidance to carry out the dry lab work.
Competing interests
The authors declare that they have no competing interests.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Funding
Not applicable.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.
About this article
Cite this article
Dhiman, P., Malik, N. & Khatkar, A. Lead optimization for promising monoamine oxidase inhibitor from eugenol for the treatment of neurological disorder: synthesis and in silico based study. BMC Chemistry 13, 38 (2019). https://doi.org/10.1186/s13065-019-0552-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1186/s13065-019-0552-4